Product Overview
 Via our Preferred Partnerships – we can help meet your recruitment goals at less cost, with exacting results and in less time – Our Comprehensive Feasibility Assessment assures recruitment goals will be met long before investing further into study launch.
Via our Preferred Partnerships – we can help meet your recruitment goals at less cost, with exacting results and in less time – Our Comprehensive Feasibility Assessment assures recruitment goals will be met long before investing further into study launch.
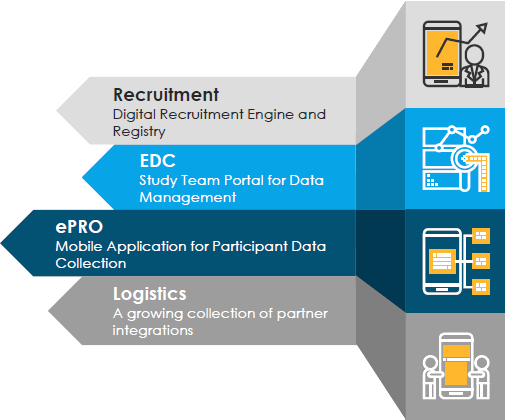
 Feasibility Testing with access to a pharmacy database of over 30 million pharmacy consumers and access to over 230 million weekly shoppers in 50 States in the U.S. within over 4000 geo-specific locations
Feasibility Testing with access to a pharmacy database of over 30 million pharmacy consumers and access to over 230 million weekly shoppers in 50 States in the U.S. within over 4000 geo-specific locations
 Our system enables Patient Identification & Engagement – fulfill enrollment requirements in weeks not years
Our system enables Patient Identification & Engagement – fulfill enrollment requirements in weeks not years
 Unprecedented Reach to the under-served populations for clinical study diversity & inclusion, for the ultimate in Health Equity
Unprecedented Reach to the under-served populations for clinical study diversity & inclusion, for the ultimate in Health Equity
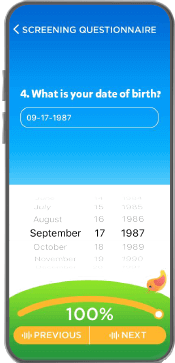
- SaaS or “Self Service” tools easily create pre-screening inclusion and exclusion criteria
- Data collection fully compatible with GCP ICH guidelines, the FDA’s 21 CFR part 11 compliance, GDPR and California’s new CCPA

- SaaS or “Self Service” tools easily create pre-screening inclusion and exclusion criteria
- Data collection fully compatible with GCP ICH guidelines, the FDA’s 21 CFR part 11 compliance, GDPR and California’s new CCPA
- Automated subject screening process – recruitment & screening information exports automatically for attachment to IRB applications
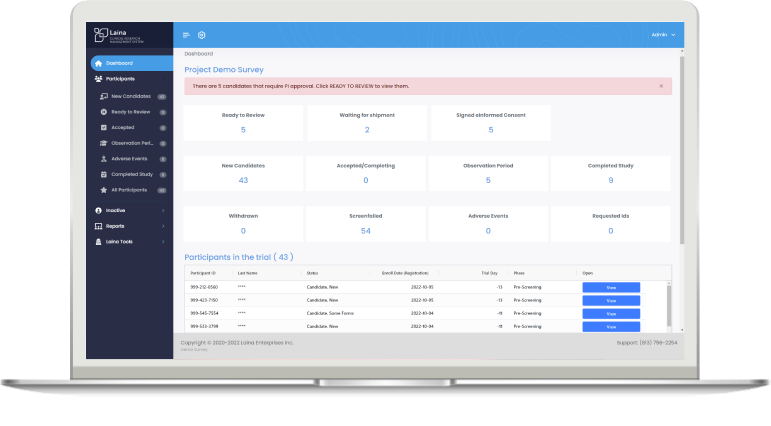
- Live Data Reporting & Digital Dashboards within our EDC, allows the Sponsor, CRO, or EA to adjust recruitment efforts in real-time
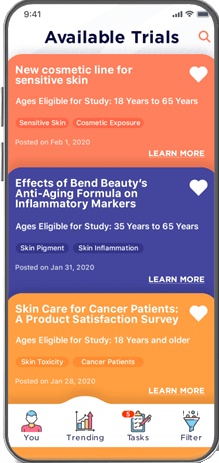
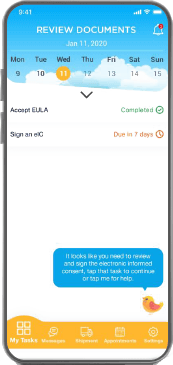
Candidates access your trials through a customized mobile app available across all popular devices and configurable in multiple languages
Designed by an experienced team to be accessible, easy to use, and highly secure
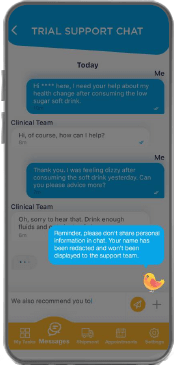
Facilitates communications with the study team and includes advanced personal information (PI) redaction


Extensively researched and tested electronic informed consent with digital source documentation and digital fingerprinting
Multiple forms of identity validation available including automatic scan of Government issue ID or passport in 150+ countries
Any documents you need, update and re-sign during the trial

01
Generate shipping information, manage tracking data, confirm through barcode scanning
02
Supports CPG, Medical Device and Drug trials and includes randomization and blinding
03
Works with our 3rd party vendors or any major shipper such as UPS, FedEx or DHL
04
Supports pharmacy-based shipping
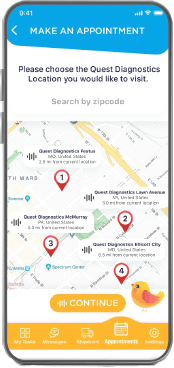
Appointment
Scheduling
ePRO
Communicate
Tasks and
Notifications
Interactive Dashboards
Rules and Role Based dynamic live data portal from the Laina Insights Engine™
- 1. Minimize drop outs
- 2. Improve protocol compliance
- 3. Optimize subject population
- 4. Enables ad hoc review of “live” blinded data
- 5. Real time TLF population allows preliminary analysis in days, not months
- 6. Supports PRO requirement for FDA submissions
- 7. Facilitates Adaptive Study Design
- Can course correct mid study for dosage, “N”, population demographics and more
- No more waiting for a study to complete that is on track to fail
- 8. Supported by Machine Learning (ML) & an Insights Engine for true Artificial Intelligence (AI)
- Important for post marketing surveillance, longitudinal data assessments and insights, as well as BYOD centric studies
- Enables the FDA’s vision for compression of the study phases
- 9. Builds the business case for Market Access via behavioral data capture
- Payor cost implications
- Utilization data
- 10. Focus:
- Tracks adherence, compliance & positive behavioral changes

Laina Enterprises Insights Engine for True AI
- Real World and Real Time data collection for Real World Evidence
- Supports new data paradigm Real World Performance
- Machine Learning (driven by custom Rules-based Engines for “Dynamic data” creating actionable insights
- The system becomes smarter over time, system intelligence pushes Targeted content/communications developed based on deeper insights
- Elimination of expensive ‘dead time’ enables the continuous study as proposed by the former FDA Commissioner Gottlieb
- Affords compression of the study phases for Accelerated implementation of Iterative studies
- Track data from individual subjects to large populations
- Globally scalable

- Company owned architecture built on open standards. No limitations, loss of ownership or control by Apple.
- Available on all major consumer platforms. Components available as needed.
- Sponsor controls the data dialogues.

Excellent for post-market surveillance
Clinically Validated easy homeuse devices collect a variety of health data

BYOD Metrics
Reliable and instant
health data

Automatic Health Data
From Apple, Fitbit, and
others
- Collect and save data without access to the Internet
- Data is transmitted when an appropriate connection is made
- Study personnel can engage participants wherever they might be to collect data
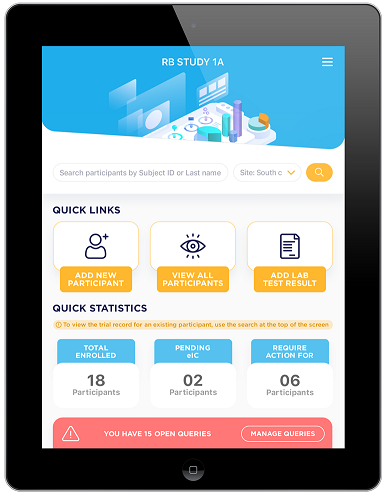
- Utilize clinically validated and ad hoc questionnaires
- Capture and respond to all lab & test data
- Record AE’s
- eConsent & eSignature
- Manage medications, vaccinations and supplementation
- Record & view Medical History including symptom tracking
- Manage daily, weekly and monthly tasks
- Capture photo data
- Query management feature
- Customizable for any use case


